Why Is the Relocation of RC School A Good and Bad Thing!
Dorian Nuñez
April 8, 2024
2 Comments
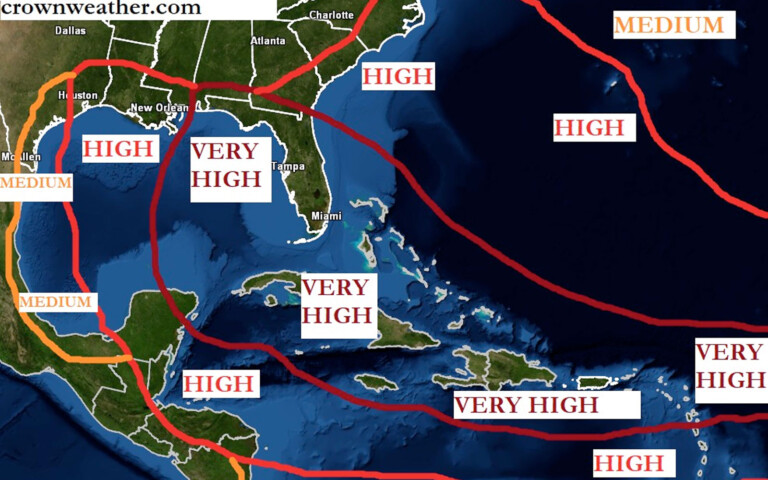
As soon as word broke that the San Pedro Roman Catholic School (RC School) was relocating from downtown San Pedro and that the property was being sold off to a developer, emotions started flying out the door and people were expressing their discontent of the move, while others are happy